 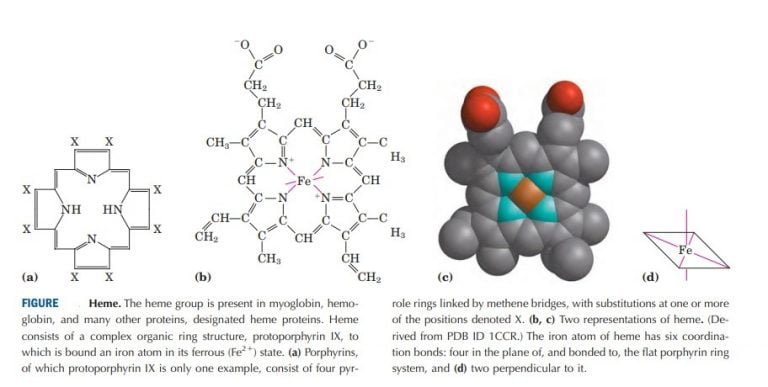
The heme group is composed of a porphyrin ring which contains an iron (Fe) atom in its center. It is composed of four subunits, each with a heme group plus a globin chain. The protein hemoglobin is a molecule which is responsible for carrying almost all of the oxygen in the blood. Oxygen is carried in two forms inside RBCs: it is dissolved in RBC water (about 70% of RBC volume is water) in accordance with Henry's law, and a much larger amount of oxygen is reversibly bound to the hemoglobin contained within the RBCs. Thus, at a P O 2 of 100 mm Hg (typical value for arterial blood), 100 ml of plasma contains 0.3 ml O 2 (or 0.3 vol%). Henry's law states that the amount of oxygen dissolved in plasma is directly proportional to P O 2: = α P O 2, where α = 0.003 ml O 2 (100 ml plasma) −1 mm Hg −1. At elevated P O 2 (breathing 100 % oxygen or during hyperbaric oxygenation), however, the physically dissolved form of oxygen can become significant. 
Oxygen is carried in the blood in two forms: (1) dissolved in plasma and RBC water (about 2% of the total) and (2) reversibly bound to hemoglobin (about 98% of the total).Īt physiological P O 2 (40 < P O 2 < 100 mm Hg), only a small amount of oxygen is dissolved in plasma since oxygen has such a low solubility. The fractional volume of blood occupied by RBCs is called the hematocrit, and its value is a little less than 50% in human adults (∼40% for females and ∼45% for males). įor purposes of discussing oxygen transport by the blood, we will consider blood to be composed of two phases: plasma and red blood cells (RBCs). This analysis can be found in many monographs on respiratory physiology, as well as the more involved case of oxygen exchange where oxygen binding to hemoglobin inside the red blood cells must be taken into account. For simplicity, gases that only exist in the physically dissolved form (e.g., He, Ar, N 2O) are usually considered, so that one does not have to deal with the complications added by binding to proteins in the blood or carriage in a chemically modified form. A similar analysis is carried out for oxygen in the peripheral circulation in Chapter 8. One can carry out an analysis of gas exchange by using Fick's first law to determine the gas transport that takes place between an alveolus and a small volume of blood as it traverses the gas exchange region of the lung. This is the case for oxygen, so that the blood flowing through the pulmonary capillaries comes into equilibrium with the P O 2 in the alveolar gas after traversing about one-third the length of the pulmonary capillaries. Gas Exchange Limited by Diffusion and PerfusionĪ quantitative description of the gas exchange characteristics of the lungs leads to the conclusion that the exchange of most gases is limited by perfusion (i.e., blood flow).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
